Blog

The Subclinical Gap: Why Mechanical Lung Function Is a Lagging Indicator in COPD
- by Manjushree Shankarappa & Hithesh K. Gatty
- 20 Feb 2026
For more than half a century, spirometry has defined how we diagnose Chronic Obstructive Pulmonary Disease (COPD). A reduced FEV_1/FVC ratio confirms airflow limitation, and clinical management follows accordingly. International guidelines continue to rely on spirometry as the diagnostic cornerstone of the disease [2].
But here is the uncomfortable truth: Spirometry measures damage. It does not measure disease activity. By the time airflow obstruction becomes measurable on a flow-volume loop, pathological processes have often been evolving for years. Small airway inflammation, oxidative stress, epithelial injury, and alveolar remodeling typically precede the mechanical decline we eventually detect [3]. This delay creates what we might call a subclinical gap–a period during which biological injury progresses while mechanical measurements remain “normal.”
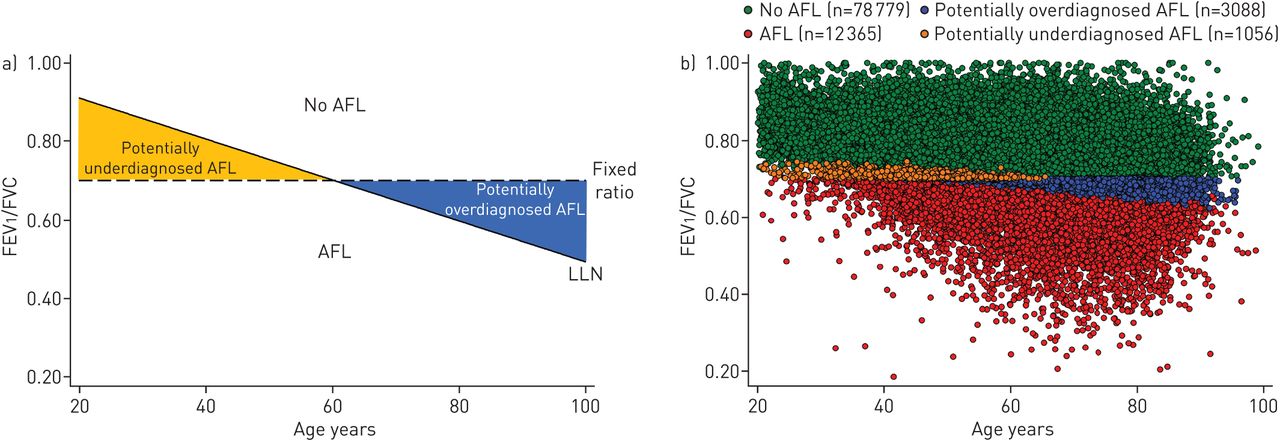
Standard spirometry often misses a “hidden” population, identified as having Preserved Ratio Impaired Spirometry (PRISm)-who exhibit significant lung impairment despite maintaining seemingly normal airflow ratios. According to the foundational study by Çolak et al. (2018), these individuals are in a dangerous “transition state,” with one in four developing full-blown COPD within five years. The limitation lies in how we measure lung health. Traditional diagnostics rely on a “fixed ratio” (0.70), which acts as a rigid, one-size-fits-all cutoff. As shown in Figure 1, this method creates a massive diagnostic gap: it leads to significant underdiagnosis in younger individuals and overdiagnosis in the elderly [1]. The implication for clinicians is clear: If COPD begins as a biological process, our monitoring strategies should reflect biology, not just mechanics.
Disease Activity vs. Damage Assessment
In many areas of medicine, we strictly distinguish between markers of damage and markers of activity.
- In Infectious Disease control, we do not wait for organ failure before measuring viral load.
- In Rheumatology, we monitor inflammatory cytokines before joint destruction becomes visible.
Yet in respiratory care, routine diagnostics still center on airflow limitation–a downstream consequence of chronic injury. This is where biochemical signals become critical.
The HO-1 Pathway: A Cellular Stress Response
Oxidative stress is central to COPD pathogenesis. Exposure to tobacco smoke, biomass fuel, and environmental pollutants induces inflammatory cascades within the airway epithelium. A key adaptive response to this assault is the upregulation of Heme Oxygenase-1 (HO-1), a cytoprotective enzyme activated during oxidative and inflammatory stress [4]. HO-1 catalyzes the degradation of heme.
The HO-1 Pathway: A Cellular Stress Response
Oxidative stress is central to COPD pathogenesis. Exposure to tobacco smoke, biomass fuel, and environmental pollutants induces inflammatory cascades within the airway epithelium. A key adaptive response to this assault is the upregulation of Heme Oxygenase-1 (HO-1), a cytoprotective enzyme activated during oxidative and inflammatory stress [4]. HO-1 catalyzes the degradation of heme. Biochemically, the reaction proceeds as follows:
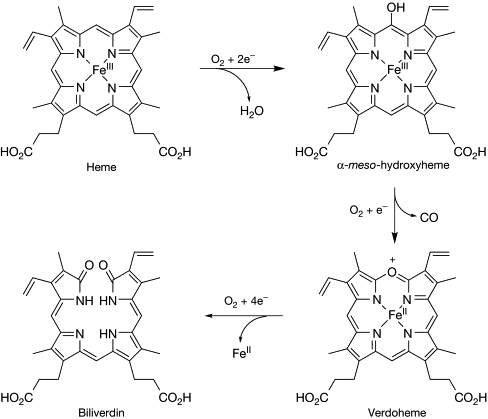
Carbon monoxide (CO), produced as a byproduct of this reaction, diffuses into the bloodstream and is exhaled. Measured in parts per million (ppm), exhaled CO (FeCO) has long been used as an objective marker of recent smoking [5]. But its relevance extends beyond behavioral validation.
Elevated FeCO levels have been consistently reported in patients with COPD compared with healthy non-smokers, reflecting the increased oxidative and inflammatory burden within the airways [6]. While not yet a standalone diagnostic, FeCO provides insight into ongoing biological stress—the type of activity that precedes structural decline.
In simple terms:
- Spirometry measures how well the lungs move air.
- Biochemical signals (FeCO) reveal how stressed the lungs are.
Both matter. But they answer different questions.
Toward Leading Indicators in COPD
If spirometry represents a lagging indicator, capturing established airflow limitation, then biochemical markers may function as leading indicators of disease activity.
This distinction has practical implications for modern primary care:
- Risk Stratification: Identifying high-risk smokers whose airflow is still borderline but whose oxidative stress levels are elevated.
- Longitudinal Monitoring: Tracking inflammatory stress over time to predict exacerbation risk before lung function crashes.
- Pharmacodynamics: Evaluating patient response to anti-inflammatory therapies (e.g., ICS or biologicals) by monitoring drops in oxidative byproducts.
Current evidence suggests that while FeCO is not a definitive predictor of all exacerbations, its biological plausibility and association with oxidative stress justify its integration into a multimodal monitoring framework [4, 6].
The goal is not to replace spirometry. The goal is to complement it.
Closing the Subclinical Gap
COPD remains a leading cause of morbidity and mortality worldwide, with a substantial socioeconomic burden driven by late diagnosis and exacerbation-related hospitalizations [7]. Shifting detection earlier requires tools that move beyond episodic damage assessment toward continuous disease activity monitoring. Portable, point-of-care technologies capable of capturing both mechanical airflow and biochemical stress signals may help narrow the subclinical gap.
The future of respiratory monitoring lies in integrating spirometry with biologically meaningful signals that reflect what is happening at the cellular level because waiting for airflow to decline is not the same as preventing it.
Reference
[1] Carpagnano, G. E., Resta, O., Foschino-Barbaro, M. P., Gramiccioni, E., & Barnes, P.J. (2003). Increased inflammatory markers in the exhaled breath condensate of cigarette smokers. European Respiratory Journal, 21(4), 589-593. https://doi.org/10.1183/09031936.03.00022203
[2] Slebos, D. J., Ryter, S. W., & Choi, A. M. K. (2003). Heme oxygenase-1 and carbon monoxide in pulmonary medicine. Respiratory Research, 4, 7. https://doi.org/10.1186/1465-9921-4-7
[3] Jarvis, M. J., Russell, M. A. H., & Saloojee, Y. (1980). Expired air carbon monoxide: A simple breath test of tobacco smoke intake. British Medical Journal (BMJ), 281(6238), 484–485. https://doi.org/10.1136/bmj.281.6238.484
[4] National Institute for Health and Care Excellence (NICE). (2008). PH10 Stop smoking services, evidence review (CO validation at 4 weeks). (PDF). https://www.nice.org.uk/guidance/ng209/evidence/ph10-stop-smoking-services-february-2008-pdf-10892313614
[5] Montuschi, P., Kharitonov, S. A., & Barnes, P. J. (2001). Exhaled carbon monoxide and nitric oxide in COPD. Chest, 120(2), 496–501. https://doi.org/10.1378/chest.120.2.496
[6] Gatty Instruments AB (2025). https://gattyinstruments.com/
[7] SPIROLUFT: A Next Generation Breath Analyser Utilising Simultaneous Spirometry & Pulse Oximetry, Säfström, Felix, Master thesis, Uppsala University, Sweden https://uu.diva-portal.org/smash/get/diva2:1916914/FULLTEXT01.pdf
[8] The Role of Heme Oxygenase-1 in Pulmonary Disease, Laura E Fredenburgh, Mark A Perrella, S. Alex Mitsialis, Division of Pulmonary and Critical Care Medicine, Department of Medicine, Brigham and Women’s Hospital; and Division of Newborn Medicine, Children’s Hospital, Boston, Massachusetts. https://www.researchgate.net/figure/Degradation-of-heme-by-HO-1-HO-1-catalyzes-heme-degradation-to-biliverdin-IX-CO-and_fig1_6812848
- Çolak, Y., et al. (2018). Characteristics and Prognosis of Individuals with Preserved Ratio Impaired Spirometry (PRISm). European Respiratory Journal, 51(4), 1702681.
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for Prevention, Diagnosis and Management of COPD: 2026 Report.
- Martinez FJ, et al. A new era for the definition and classification of COPD: Pre-COPD and PRISm. Eur Respir J. 2022.
- Ryter SW, Alam J, Choi AMK. Heme oxygenase-1/carbon monoxide: from basic science to therapeutic applications. Physiol Rev. 2006;86:583-650.
- SRNT Subcommittee on Biochemical Verification. Biochemical verification of tobacco use and cessation. Nicotine Tob Res. 2002;4:149-159.
- Paredi P, et al. Exhaled carbon monoxide levels in patients with COPD. Eur Respir J. 2000;15:755-760.
- World Health Organization. Chronic obstructive pulmonary disease (COPD) fact sheets and global burden data. 2025.


