Blog

Cardiovascular Risk After an Acute COPD Exacerbation (AECOPD), Why Early Detection Matters
- by Mohamed Al-Sabri & Hithesh K. Gatty
- 19 Feb 2026
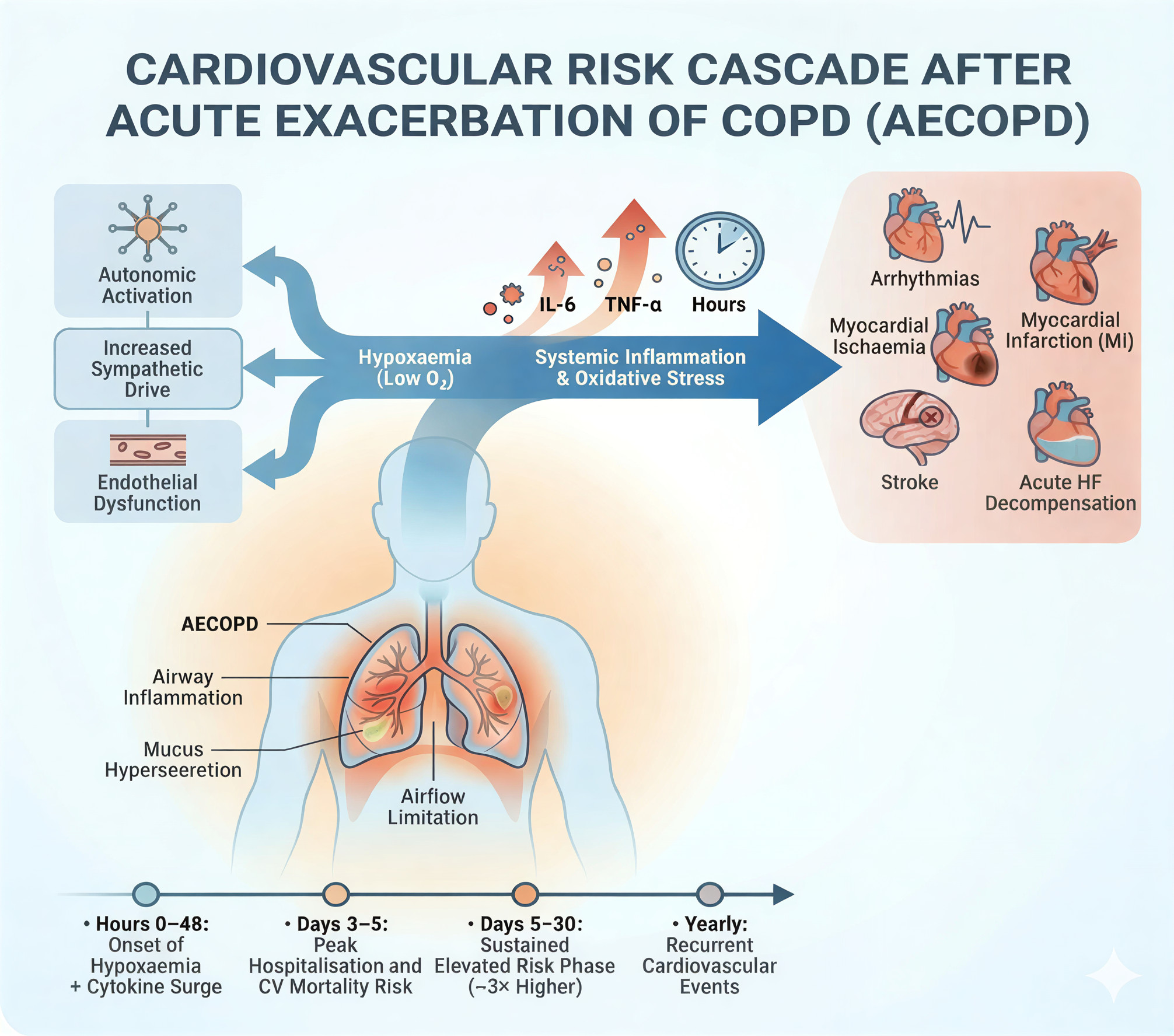
An acute exacerbation of COPD (AECOPD) is typically framed as a lung event, worsening breathlessness, cough, or sputum that prompts additional therapy. What is often under-recognised is that an exacerbation can also act as a systemic cardiovascular stressor. In the hours and days after onset, there is a short, high-risk window in which acute cardiovascular events become more likely, including myocardial infarction, arrhythmias, acute heart-failure decompensation, and stroke [1–3].
Quick Takeaways
- The highest relative risk appears immediately after AECOPD onset, particularly in the first week [2,4].
- Risk elevation is not limited to severe (hospitalised) exacerbations, moderate exacerbations also show increased cardiovascular risk [4].
- GOLD highlights increased cardiovascular-event risk during, and for at least 90 days after, acute COPD exacerbations [1].
- Earlier recognition of AECOPD supports timely respiratory escalation and prompts structured cardiovascular vigilance during the danger window [1,4].
The Danger Window After AECOPD
Multiple study designs converge on the same message: cardiovascular risk rises promptly after AECOPD onset and is greatest early.
In a self-controlled case series, the risk of myocardial infarction was about doubled in the first 1 to 5 days after exacerbation onset, and stroke risk was also increased in the post-exacerbation period [2].
More recently, a meta-analysis of the EXACOS-CV studies reported a roughly tenfold increased risk of severe cardiovascular event or death in days 1 to 7 following the onset of an exacerbation of any severity (pooled HR 10.22, 95% CI 5.34–19.57), with risk remaining elevated for up to one year [4].
A systematic review and meta-analysis similarly concluded that COPD exacerbations are associated with a markedly increased short-term risk of acute myocardial infarction and stroke [3].
Why an Exacerbation Can Trigger Cardiovascular Events
The link between AECOPD and acute cardiovascular events is biologically plausible and clinically consistent. Key pathways include:
- Hypoxaemia and respiratory strain. Lower oxygenation and increased work of breathing can promote myocardial oxygen supply-demand mismatch, especially in people with limited cardiovascular reserve.
- Systemic inflammation and oxidative stress. Exacerbations can be accompanied by systemic inflammatory responses that may destabilise atherosclerotic plaques, promote endothelial dysfunction, and increase thrombotic potential [3,6].
- Autonomic imbalance and arrhythmia vulnerability. Acute illness can increase sympathetic drive and reduce physiological stability, raising vulnerability to arrhythmias and decompensation [5].
- Comorbidity interactions. Cardiovascular disease is common in COPD, and exacerbation stress can unmask or worsen coronary disease and heart failure [1,5].
IL-6, TNF-α, and Exhaled CO, What is The Link?
Putting this together, eCO is best viewed as a broad marker of oxidative stress and inflammation, not a specific readout of IL‑6 or TNF‑α alone. However, in an AECOPD context, serial eCO monitoring may still be useful as one component of earlier recognition of inflammatory escalation, particularly in non-smokers or after controlling for smoking exposure. Earlier detection of AECOPD can support timely respiratory escalation and prompt cardiovascular vigilance during the danger window, potentially enabling earlier intervention before decompensation [1,4].
In COPD exacerbations specifically, eCO has been investigated as a practical breath measurement during severe AECOPD requiring hospitalization, including evaluation of whether monitoring changes in eCO could aid assessment during treatment and stabilisation [13].
Human data supporting an IL‑6 to eCO association exist. In cigarette smokers, IL‑6 measured in exhaled breath condensate was higher than in non-smokers and was reported to relate to exhaled carbon monoxide and lung function metrics, supporting the idea that heightened airway inflammatory activity can align with higher eCO signals [9]. A critical limitation is that, in current smokers, exogenous CO from tobacco can dominate eCO measurements, so smoking exposure must be controlled when interpreting eCO as an inflammation proxy [8,9].
Exhaled carbon monoxide (eCO) connects to this biology through the heme oxygenase (HO) pathway. Endogenous carbon monoxide is produced when heme is degraded by HO, particularly the inducible stress‑response isoform HO‑1, which is upregulated by oxidative stress and inflammatory signalling [8]. Experimental work has also shown that inflammatory cytokines including TNF‑α can induce HO‑1 expression in human vascular endothelial cells, providing a mechanistic basis for increased endogenous CO generation during inflammatory stress [10].
During an acute exacerbation of COPD (AECOPD), systemic inflammation and oxidative stress can rise rapidly. Cytokines such as interleukin‑6 (IL‑6) and tumour necrosis factor‑α (TNF‑α) are frequently discussed in this context, because exacerbations are associated with measurable systemic inflammatory responses, including elevated IL‑6 during hospital treatment and recovery periods [11,12]. This matters clinically because AECOPD is followed by a short high‑risk window for cardiovascular events, and inflammation and oxidative stress are plausible contributors to endothelial dysfunction, thrombogenicity, arrhythmia vulnerability, and plaque instability during that period [2-6].
Who is at Higher Risk?
Risk is often higher in people with prior cardiovascular disease, older age, higher comorbidity burden, and those experiencing severe or recurrent exacerbations [4,5]. The key practical point is that AECOPD is common, so even moderate relative risk increases can translate into substantial clinical burden.
Why Early Detection Changes the Prevention Logic
If the highest hazard is concentrated in the first days after AECOPD onset, timing of recognition becomes critical. In many care pathways, exacerbations are detected late, when symptoms have already escalated to emergency care. Earlier recognition of impending or early AECOPD can enable timely respiratory treatment escalation and monitoring, and can also trigger structured cardiovascular vigilance during the danger window, such as triage for chest pain or palpitations, ECG when indicated, and review of heart-failure symptoms [1,4].
This is not about turning COPD care into cardiology follow-up, it is about acknowledging that AECOPD is a predictable cardiopulmonary risk state and acting early to reduce avoidable deterioration.
Figure
Reference
[1] Carpagnano, G. E., Resta, O., Foschino-Barbaro, M. P., Gramiccioni, E., & Barnes, P.J. (2003). Increased inflammatory markers in the exhaled breath condensate of cigarette smokers. European Respiratory Journal, 21(4), 589-593. https://doi.org/10.1183/09031936.03.00022203
[2] Slebos, D. J., Ryter, S. W., & Choi, A. M. K. (2003). Heme oxygenase-1 and carbon monoxide in pulmonary medicine. Respiratory Research, 4, 7. https://doi.org/10.1186/1465-9921-4-7
[3] Jarvis, M. J., Russell, M. A. H., & Saloojee, Y. (1980). Expired air carbon monoxide: A simple breath test of tobacco smoke intake. British Medical Journal (BMJ), 281(6238), 484–485. https://doi.org/10.1136/bmj.281.6238.484
[4] National Institute for Health and Care Excellence (NICE). (2008). PH10 Stop smoking services, evidence review (CO validation at 4 weeks). (PDF). https://www.nice.org.uk/guidance/ng209/evidence/ph10-stop-smoking-services-february-2008-pdf-10892313614
[5] Montuschi, P., Kharitonov, S. A., & Barnes, P. J. (2001). Exhaled carbon monoxide and nitric oxide in COPD. Chest, 120(2), 496–501. https://doi.org/10.1378/chest.120.2.496
[6] Gatty Instruments AB (2025). https://gattyinstruments.com/
[7] SPIROLUFT: A Next Generation Breath Analyser Utilising Simultaneous Spirometry & Pulse Oximetry, Säfström, Felix, Master thesis, Uppsala University, Sweden https://uu.diva-portal.org/smash/get/diva2:1916914/FULLTEXT01.pdf
[8] The Role of Heme Oxygenase-1 in Pulmonary Disease, Laura E Fredenburgh, Mark A Perrella, S. Alex Mitsialis, Division of Pulmonary and Critical Care Medicine, Department of Medicine, Brigham and Women’s Hospital; and Division of Newborn Medicine, Children’s Hospital, Boston, Massachusetts. https://www.researchgate.net/figure/Degradation-of-heme-by-HO-1-HO-1-catalyzes-heme-degradation-to-biliverdin-IX-CO-and_fig1_6812848
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of COPD, 2025 Report (v1.0, 15 Nov 2024). https://goldcopd.org/wp-content/uploads/2024/11/GOLD-2025-Report-v1.0-15Nov2024_WMV.pdf
- Donaldson GC, Hurst JR, Smith CJ, Hubbard RB,WedzichaJA. Increased risk of myocardial infarction and stroke following exacerbation of COPD. Chest. 2010. PubMed: https://pubmed.ncbi.nlm.nih.gov/20022970/
- Müllerová H, Marshall J, de Nigris E, et al. Association of COPD exacerbations and acute cardiovascular events: a systematic review and meta-analysis. Therapeutic Advances in Respiratory Disease. 2022. DOI: https://doi.org/10.1177/17534666221113647; PubMed: https://pubmed.ncbi.nlm.nih.gov/35894441/; Full text (PMC): https://pmc.ncbi.nlm.nih.gov/articles/PMC9340406/
- Nordon C, Simons SO, Marshall J, et al. The sustained increase of cardiovascular risk following COPD exacerbations: meta-analysesof the EXACOS-CV studies. ERJ Open Research. 2025 (online first 2024). PubMed: https://pubmed.ncbi.nlm.nih.gov/40524923/; Full text (PMC): https://pmc.ncbi.nlm.nih.gov/articles/PMC12168178/; Full PDF: https://publications.ersnet.org/content/erjor/11/3/01091-2024.full.pdf
- Reilev M, Pottegård A, Lykkegaard J, et al. Increased risk of major adverse cardiac events following the onset of acute exacerbations of COPD. Respirology. 2019. PubMed: https://pubmed.ncbi.nlm.nih.gov/31222861/
- Swart KMA, Broekhuizen BDL, Gucht J, et al. Risk of cardiovascular events after an exacerbation of chronic obstructive pulmonary disease. Respiratory Research. 2023. PDF: https://respiratory-research.biomedcentral.com/counter/pdf/10.1186/s12931-023-02601-4.pdf
- European Society of Cardiology (ESC)CardioPractice. Cardiovascular risk in chronic obstructive pulmonary disease. 2025. https://www.escardio.org/communities/councils/cardiology-practice/education/cardiopractice/cardiovascular-risk-in-chronic-obstructive-pulmonary-disease/
- Ryter SW, Choi AMK. Carbon monoxide in exhaled breath testing and therapeutics. Journal of Breath Research. 2013;7(1):017111. DOI: https://doi.org/10.1088/1752-7155/7/1/017111; PubMed: https://pubmed.ncbi.nlm.nih.gov/23446063/; Full text (PMC): https://pmc.ncbi.nlm.nih.gov/articles/PMC3651886/
- CarpagnanoGE, Kharitonov SA, Foschino-Barbaro MP, Resta O, Gramiccioni E, Barnes PJ. Increased inflammatory markers in the exhaled breath condensate of cigarette smokers. European Respiratory Journal. 2003;21(4):589–593. DOI: https://doi.org/10.1183/09031936.03.00022203; PubMed: https://pubmed.ncbi.nlm.nih.gov/12762340/; ERS PDF: https://publications.ersnet.org/content/erj/21/4/589.full.pdf
- Terry CM,ClikemanJA, Hoidal JR, Callahan KS. TNF‑α and IL‑1α induce heme oxygenase‑1 via protein kinase C, phospholipase A2, and oxidant-dependent mechanisms in endothelial cells. American Journal of Physiology-Heart and Circulatory Physiology. 1999;276(5):H1493–H1501. DOI: https://doi.org/10.1152/ajpheart.1999.276.5.H1493; PubMed: https://pubmed.ncbi.nlm.nih.gov/10330231/
- Groenewegen KH,DentenerMA, Wouters EFM. Longitudinal follow-up of systemic inflammation after acute exacerbations of COPD. Respiratory Medicine. 2007. PubMed: https://pubmed.ncbi.nlm.nih.gov/17644367/; Full text: https://www.resmedjournal.com/article/S0954-6111%2807%2900259-4/fulltext
- Wouters EFM, Groenewegen KH,DentenerMA, Vernooy JHJ. Systemic inflammation in chronic obstructive pulmonary disease: the role of exacerbations. Proceedings of the American Thoracic Society. 2007;4(8):626–634. DOI: https://doi.org/10.1513/pats.200706-071TH; PubMed: https://pubmed.ncbi.nlm.nih.gov/18073394/
- Antus B,DrozdovszkyO, Barta I. Assessment of exhaled carbon monoxide in exacerbations of chronic obstructive pulmonary disease. Physiology International. 2016;103(2). DOI: https://doi.org/10.1556/036.103.2016.2.8; PubMed: https://pubmed.ncbi.nlm.nih.gov/28639864/; PDF: https://real.mtak.hu/38094/1/036.103.2016.2.8.pdf


