Our Products

Advanced Respiratory Analyser
Breathe smarter with a single portable device that measures Spirometry and FECO, empowering early detection and personalized lung care.
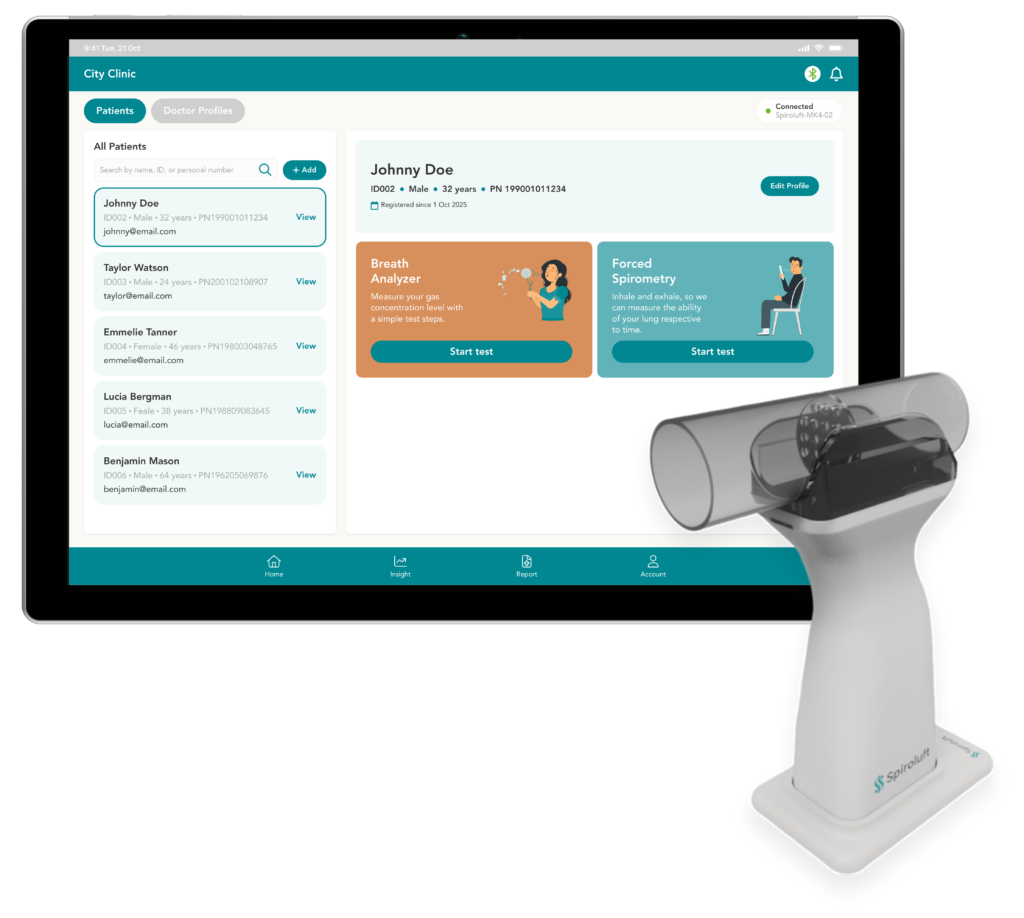
Technical Specifications
Key specifications and performance metrics supporting reliable clinical use.
FEco (Fractional Exhaled Carbon Monoxide) | ±0.1ppm (parts per million) |
Spirometer accuracy | ±2.5% or ±0.05 L (whichever is greater) |
Dimensions | 150mm x 70mm x 40mm |
Weight | 250g |
Display | Versatile mobile application |
Battery life | Up to 12 hours of continuous use |
Data storage | Cloud-based environment |
Connectivity | Bluetooth, USB |
Upcoming Products:
Volumetric Capnography
More info coming soon.

Advanced Respiratory Analyser
Breathe smarter with a single portable device that measures Spirometry, FECO, and Respiratory Rate, empowering early detection and personalized lung care.*


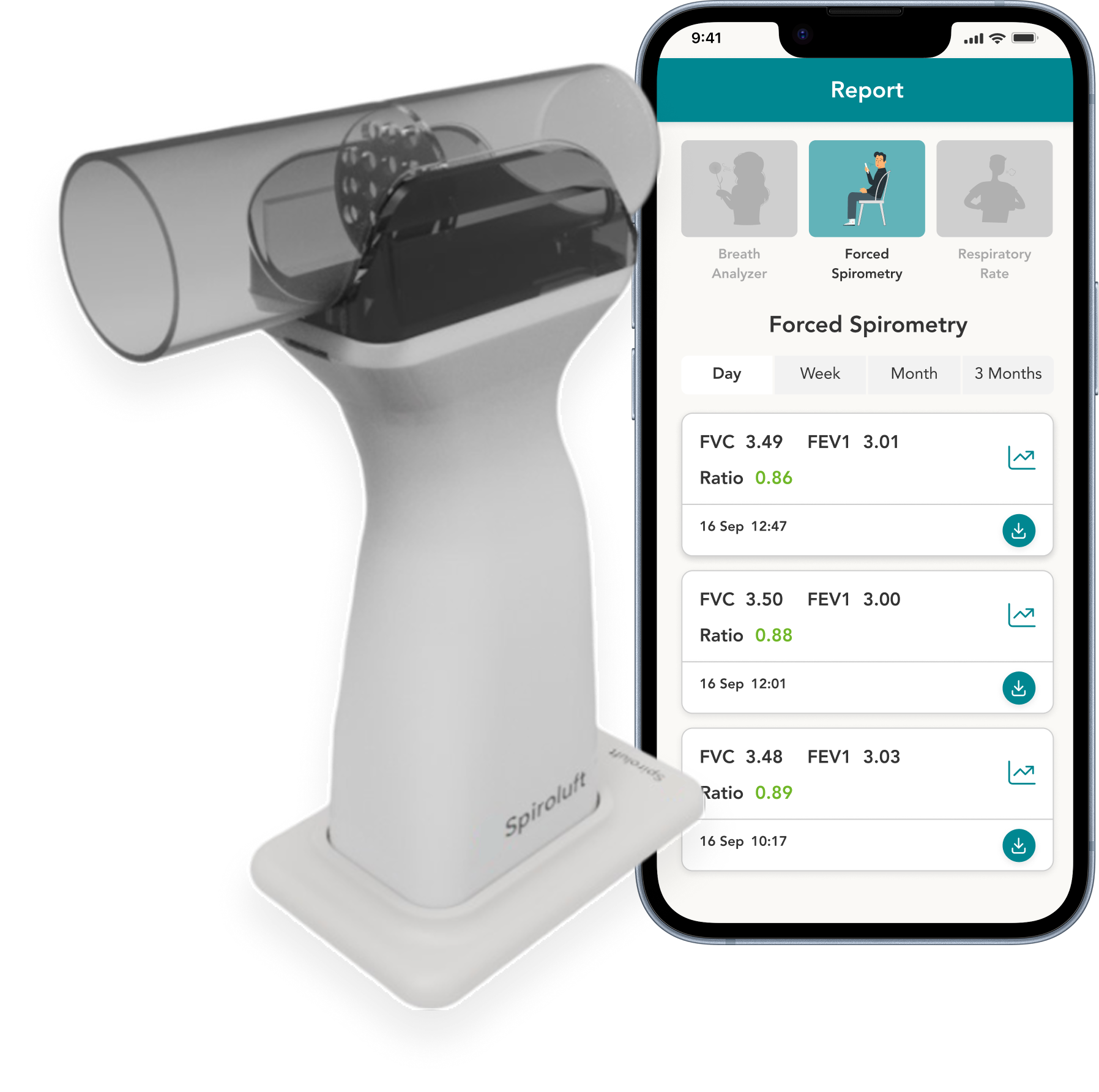
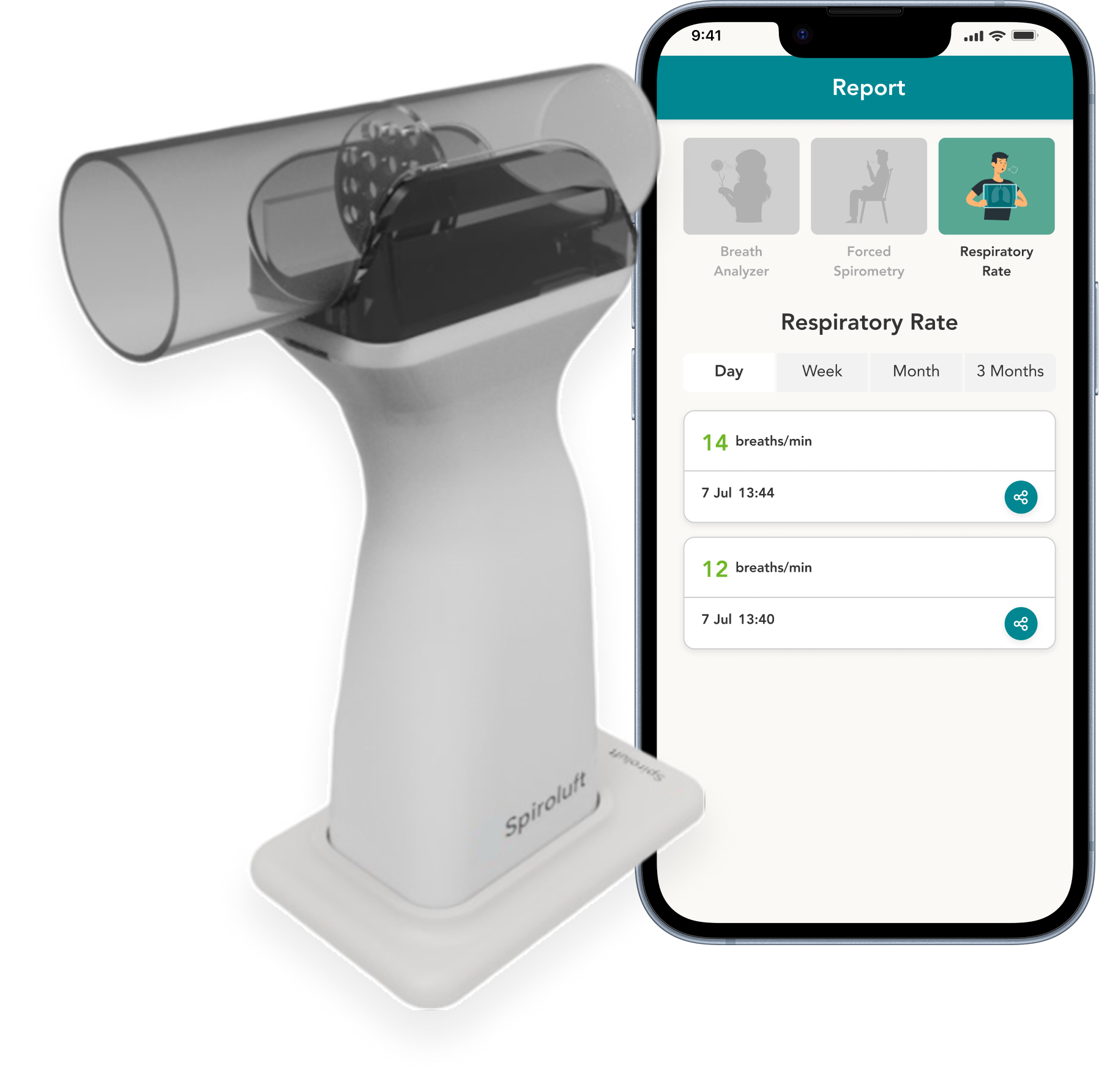

Device
- Device Dimensions: 150 X 70 X 40 mm
- Weight: 250 grams
Electrical Data
- Power: Battery operation
- Power Cable: USB A to C
- Power Consumption: 0.56 Watt (Approx.)
- Battery: Rechargeable Lithium Polymer Battery,
3.7 V, 2000 mAh - Battery Capacity: 12 to 13 Hrs Normal use
Mouthpiece
Disposable, OD 30 mm, resistance (cm H2O) <1.3@60L/min with bacterial filtration 99.99%
Interfaces
- BLE Communication
- Recording Functions
- Measurements: FECO, FVC, FEV1, PEF & RR Measurement
- Standard Spirometry: ATS ERS 2005/2019 update, GLI 2012
- Measurement Standards: GOLD
Memory
Cloud based solution
Spiroluft
- Measurement Method: Dual-Sensor Technology
- Measurement Range: Flow 0 to ±16 L/s
- Volume: No Limit
- Measurement Accuracy: Flow ±2.5% or 25 ml/s &
Volume ±2% or 50 ml - Temperature Drift: Max ±2%
- Respiratory Resistance: Less than 0.15 kPa/L/s
- Standards Compliance: ATS
Environmental Conditions Sensor
The Environment conditions for the sensor depend on the user operations and are determined by it.
Temperature
- Operating: +15 °C to +50 °C
- Transport/Storage: -20 °C to +50 °C
Relative Humidity
- Operating: 10 to 95% RH, non-condensing
- Transport/Storage: 10 to 95% RH, non-condensing
Standards
Certification: Undergoing Class according to IEC60601-1:I

Advanced Respiratory Analyser
Breathe smarter with a single portable device that measures Spirometry, FECO, and Respiratory Rate, empowering early detection and personalized lung care.*




